Unidirectional Air Flow
Ensuring contamination-free environments with precise airflow control for cleanroom and high-grade sterile applications.
Unidirectional Air Flow
When a Unidirectional Air Flow (UDAF) system is connected to an Air Handling Unit (AHU) and used as a single pass-through system, it typically refers to a setup commonly found in controlled environments such as cleanrooms, laboratories, or certain manufacturing injectable aseptic filling processes
Unidirectional Air Flow (UDAF) System:
It creates a controlled airflow pattern where air moves in a single direction within a defined space. It maintains a clean environment over a filling line for pharmaceutical manufacturing by removing particles and contaminants.
Air Handling Unit (AHU):
An AHU conditions, filters, and circulates air within a building or controlled environment. In this context, the AHU provides the filtered and conditioned air that the UDAF system utilizes. It ensures the quality of the air supplied to the UDAF and subsequently to the filling line.

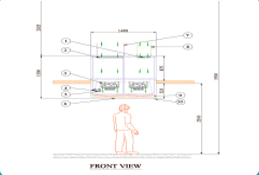
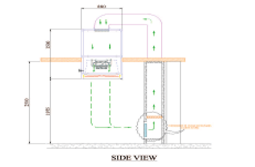
Single Pass-Through System:
The air passes through the UDAF and the controlled environment only once before being exhausted or possibly recirculated. This setup is used when maintaining a specific level of cleanliness is necessary and recirculating the air might carry contaminants back into the controlled environment.
Building Management System (BMS):
It is a centralized system that monitors and controls various building functions, including HVAC (Heating, Ventilation, and Air Conditioning) systems. In your case, the BMS controls the AHU and possibly the UDAF system as well. This helps to keep optimal conditions, efficiency, and coordination between different systems in the facility
In summary, your described scenario involves using a Unidirectional Air Flow (UDAF) system connected to an Air Handling Unit (AHU) in a single pass-through system over a filling line. A Building Management System (BMS) controls and coordinates this setup to maintain a clean and controlled environment crucial for sensitive processes. This method ensures the safety and quality of products in pharmaceutical or electronics manufacturing

DOWNLOAD BROCHURE (PDF)
Unidirectional Air Flow: The Backbone of Sterile Cleanroom Environments
Introduction
In highly sensitive environments like pharmaceutical manufacturing, biotechnology labs, healthcare facilities, and electronics production, contamination control is non-negotiable. At the heart of contamination control lies a critical engineering solution: Unidirectional Air Flow. This technology forms the core of controlled cleanroom environments where cleanliness, sterility, and compliance with global standards are essential.
In this blog, we will explore the significance of Unidirectional Air Flow, dive into its implementation through Laminar Air Flow for Cleanrooms, examine the role of Vertical Laminar Air Flow Units, and explain how it aligns with GMP-Compliant Air Flow Solutions. If you’re planning a sterile facility or cleanroom upgrade, understanding how Cleanroom Unidirectional Air Flow works could save your business time, cost, and compliance headaches.
1. What Is Unidirectional Air Flow?
Unidirectional Air Flow refers to the movement of filtered air in a single, uniform direction—either vertically or horizontally. This continuous flow helps remove airborne particles and microorganisms by sweeping them out of the sterile zone.
Key Characteristics:
Consistent velocity (typically 0.3–0.45 m/s)
Minimal turbulence
Uses HEPA or ULPA filters
Achieves ISO Class 5–8 standards depending on configuration
Unidirectional air flow is essential for applications that require particle-free, microorganism-controlled air—especially in sterile manufacturing and hospital operating environments.
2. Importance of Laminar Air Flow for Cleanrooms
Laminar Air Flow for Cleanrooms is the most common application of unidirectional airflow. This system creates a protective environment by directing clean air uniformly across workspaces, typically in vertical or horizontal streams.
Benefits of Laminar Air Flow for Cleanrooms:
Reduces risk of cross-contamination
Maintains air quality in ISO and GMP-compliant zones
Creates low-turbulence, high-purity airflow
Ideal for sterile filling, weighing, sampling, and testing
In cleanrooms, Laminar Air Flow for Cleanrooms is integrated into ceiling modules, benches, or standalone cabinets. This system is especially useful for aseptic processing where microbial control is mission-critical.
3. Vertical Laminar Air Flow Units: Structure & Application
Among the various configurations, Vertical Laminar Air Flow Units are the most popular in cleanroom design. These systems direct air from the ceiling vertically downward over the workspace and out through floor-level or wall exhaust grills.
Key Features:
Ceiling-mounted HEPA filters
Low-noise centrifugal fans
Stainless steel or powder-coated casings
Vibration-free construction
Optional lighting and UV sterilization
Applications of Vertical Laminar Air Flow Units:
Aseptic filling lines
Injectable vial/bottle filling
Biosafety cabinets
Modular operating theatres
Research & microbiology labs
These Vertical Laminar Air Flow Units offer flexibility, reduced footprint, and exceptional contamination control—making them ideal for Class 100 (ISO 5) environments.
4. Cleanroom Unidirectional Air Flow: How It Works
Cleanroom Unidirectional Air Flow systems are typically integrated into the cleanroom ceiling or walls. The system provides air changes per hour (ACH) that exceed 300–600 in sterile zones—ensuring constant dilution and removal of particles.
Working Mechanism:
Air enters through HEPA filters at the ceiling level.
Uniform velocity air flows downward (vertical laminar) or across (horizontal laminar).
Particles and contaminants are swept toward return air ducts or floor-level exhaust grills.
Air recirculates or is replaced with fresh, filtered air.
This uninterrupted flow of clean air drastically reduces airborne contaminants and supports GMP-Compliant Air Flow Solutions for pharmaceutical production, compounding areas, and clean surgical zones.
5. Meeting Global Standards with GMP-Compliant Air Flow Solutions
In pharmaceutical and healthcare sectors, following GMP-Compliant Air Flow Solutions is a legal and regulatory requirement. Cleanroom airflow systems must be validated and documented to ensure that they conform to standards like:
WHO GMP
USFDA cGMP
EU GMP Annex 1
ISO 14644 Cleanroom Standards
To comply with these, your Unidirectional Air Flow system must:
Be equipped with validated HEPA filters
Maintain differential pressure between cleanroom zones
Have continuous monitoring for temperature, humidity, and particle counts
Undergo DQ, IQ, OQ, and PQ validation stages
A poorly designed airflow system can result in regulatory non-compliance, batch contamination, or audit failures. Hence, GMP-Compliant Air Flow Solutions must be engineered and tested by experienced cleanroom consultants.
6. Key Industries Using Unidirectional Air Flow Systems
a) Pharmaceuticals
Sterile injectable lines
OSD manufacturing rooms
Sampling & dispensing booths
b) Biotech and Life Sciences
Cell culture rooms
Tissue engineering labs
c) Hospitals and Modular Operation Theatres
Operating theatres (modular OT)
ICU and isolation rooms
d) Semiconductors & Electronics
Wafer fabrication
Precision assembly
These sectors require Cleanroom Unidirectional Air Flow systems to ensure that both product integrity and personnel safety are never compromised.
7. Common Types of Laminar Flow Equipment
When implementing Laminar Air Flow for Cleanrooms, several equipment types are available:
Vertical Laminar Air Flow Units – Ceiling to floor airflow
Horizontal Laminar Air Flow Cabinets – Back-to-front airflow
Bio-Safety Cabinets (Class I, II, III) – Air containment and filtration
Sampling Booths – Downward laminar airflow for raw material handling
Laminar Benches – Localized air protection for sensitive operations
Each piece of equipment is designed to achieve unidirectional flow, prevent contamination, and maintain aseptic conditions across different cleanroom processes.
8. Installation & Validation of Unidirectional Air Flow Systems
Proper installation and validation of Unidirectional Air Flow systems are crucial for their performance.
Installation Best Practices:
Position filters and return grills to maintain air balance
Ensure room pressurization hierarchy
Avoid turbulent zones caused by equipment or furniture
Incorporate easily accessible maintenance panels
Validation Includes:
Airflow visualization (smoke test)
Velocity measurements
Particle count tests
HEPA filter integrity test (DOP test)
Temperature and humidity control checks
Documentation from these validation processes supports GMP-Compliant Air Flow Solutions and helps pass regulatory audits.
9. Maintenance of Cleanroom Unidirectional Air Flow Systems
To ensure optimal performance, Cleanroom Unidirectional Air Flow systems must be regularly maintained.
Maintenance Checklist:
HEPA filter replacement every 1–3 years
Daily airflow velocity checks in critical zones
Calibration of sensors and alarm systems
Fan and motor inspections
Cleanroom cleaning protocols
A proactive maintenance schedule reduces downtime, extends equipment life, and maintains compliance with global cleanroom standards.
10. Cost Considerations and ROI
While Laminar Air Flow for Cleanrooms and Vertical Laminar Air Flow Units can have high upfront costs, the long-term ROI is substantial:
Lower product rejection rates
Reduced contamination risks
Faster audit clearances
Extended facility lifespan
Brand reputation in regulated markets
Investing in GMP-Compliant Air Flow Solutions is not just about compliance—it’s a business decision that safeguards productivity and customer trust.
11. Case Study: Laminar Air Flow for a Sterile Injectable Plant
Client: WHO-GMP certified pharma company
Location: Maharashtra, India
Scope: Design & install cleanroom with Unidirectional Air Flow and Vertical Laminar Air Flow Units
Deliverables:
HVAC with HEPA filters (ISO Class 5)
Modular cleanroom panels
Laminar sampling booths
18 validation reports for GMP documentation
Outcome:
Passed WHO inspection on first attempt
Achieved 30% increase in operational throughput
Zero microbial contamination for 8 months
This highlights the importance of quality engineering and validated Cleanroom Unidirectional Air Flow systems in mission-critical environments.
12. Why Choose Us for Your Cleanroom Air Flow Solutions
We specialize in delivering turnkey, validated, and GMP-Compliant Air Flow Solutions for clients in pharma, biotech, hospitals, and semiconductors.
What Sets Us Apart:
15+ years of cleanroom design experience
In-house validation and HVAC engineering teams
Custom solutions for vertical or horizontal airflow
Regulatory audit support
Cost-effective AMC and maintenance plans
Our experts ensure that every Unidirectional Air Flow project meets international standards and exceeds client expectations.
Conclusion: Power Your Cleanroom with Proven Air Flow Technology
Unidirectional Air Flow is the invisible guardian of product safety, process efficiency, and regulatory compliance in controlled environments. Whether you need Laminar Air Flow for Cleanrooms, custom Vertical Laminar Air Flow Units, or an end-to-end GMP-Compliant Air Flow Solution, choosing the right technology and team makes all the difference.
📞 Call +91 9967436808 today to get expert guidance on designing, installing, and validating your next Cleanroom Unidirectional Air Flow system.
Table of Contents
Toggle